Applications of Ion Chromatography in the Analysis of Pharmaceutical and Biological Products
Here, we shall discuss application of ion chromatographic methods in the analysis of therapeutic products and their ingredients included in USP-NF monographs and general chapters, and their importance in setting standards for identity, strength content , quality and purity of products and ingredients. For more in depth understanding of the principles, instrumentation and applications of IC, readers are encouraged to read the books and other publications on IC that provide in-depth review of this chromatographic technique [].
Monographs of products and their ingredients active and inactive components include definitions or description of the articles, packaging, storage and other requirements, and specifications. The specification consists of a series of tests, one or more analytical procedure for each test, and the acceptance criteria for each procedure. General chapters provide procedures, sometimes with acceptance criteria and other requirements, in order to compile repetitive information in one location, which would otherwise appear in many monographs. In most of the other countries, pharmacopeia is part of the government.
However, in the United States, the pharmacopeia USP is a non-government not-for-profit organization that supports itself from the sale of books USP-NF and other publications and reference standards. It does not receive any financial support from federal, state or local governments or non-government organizations.
USP-NF standards are recognized not only in the Unites States but also in many other countries because they are authoritative, science-based, and are established through a transparent and credible process with established integrity. The transparency and credibility of the monographs come through the open review and comment process that takes place when proposed monographs are published in the Pharmacopeial Forum PF.
Although the Council of Experts of USP is the ultimate decision making body for the USP-NF standards, these standards are developed through public involvement and substantial interaction between USP and the stakeholders through consensus building. A stakeholder may be an institution or individual, domestic or international. Interested and knowledgeable stakeholders can provide scientific and regulatory comments to monographs published in PF. The public participation in the monograph development and revision process results in consensus among many individuals and groups, including scientific and trade organizations.
The members of the Expert Committees are unpaid volunteers who are experts in their respective fields and who participate in the USP process as individual scientists and not as representatives of their employers or any trade association, thereby eliminating the conflict of interest and providing unbiased authoritative and science-based standards.
A manufacturer sponsor submits a proposal for a new monograph or a revision to an existing monograph to USP Request for Revision.

The information and data that need to be provided with a Request for Revision is available at the USP website www. USP staff review the proposals for the appropriateness and completeness, and, when satisfied, publish the proposal to PF as In-process Revision for public comment.
After the public comment period, members of an appropriate Expert Committee together called Council of Experts reviews the proposal and the comments and approves or disapproves the request for official adoption. However, it should be noted that the manufacturers are not required at any stage to submit proposals for a new monograph or a revision to an existing monograph.
Such submission is discretionary to the manufacturers and they do so voluntarily to support the mission of the development of consensus standards for therapeutic products. The IC involves separation based on ionic interactions between ionic or polar analytes, ions present in the eluent, and ionic functional groups derivatized to the chromatographic support.
This can lead to two distinct mechanisms of separation— a ion exchange due to competitive ionic binding attraction to the chromatographic support column resin , and b ion exclusion due to repulsion between similarly charged analyte ions and the ions derivatized on the chromatographic support. Separation based on ion exchange has been used for a long time and is the predominant form of IC to-date.
However, increasing applications of ion exclusion chromatography have been reported more recently. In addition, chromatographic methods in which the separation due to ion exchange or ion exclusion is modified by the hydrophobicity of the analytes and the chromatographic support materials, presence of the organic modifiers in the eluent or due to ion-pair agents, resulting in better resolution of analytes or separation that were not achieved before, have gained popularity recently due to increased applications of mixed mode columns. Ion-exchange chromatography involves separation of ionic and polar analytes on a chromatographic support, which is derivatized with ionic functional groups that have charges opposite that of the analyte ions.
Thus, a column used to separate cations, called a cation-exchange column, contains negative ions. Similarly, an anion-exchange column, which separates anions, is derivatized with positively charged ions. The separation is effected by repeated binding of the analyte ion to the ionic sites on the chromatographic support and desorption by the ions present in the mobile phase. The ion-exchange method of separation is widely used in the analysis of anions and cations, including metal ions, mono- and oligosaccharides, sugar alcohols and other polyhydroxy compounds, aminoglycosides antibiotics , amino acids and peptides, organic acids, amines, alcohols, phenols, thiols, nucleotides and nucleosides, and other polar molecules.
Ion-exclusion Chromatography uses strong cation- or anion-exchange chromatographic supports to separate ionic analytes from polar, weakly polar and neutral analytes, and has been used typically in the analysis of organic acids, alcohols, glycols, sugars, and other weakly polar compounds. In contrast to the ion-exchange chromatography, the charge on the functional groups on the chromatographic support is same as the charge on the analyte ion.
That is, to separate negatively charged or negatively polarized analytes, the chromatographic supports are derivatized with negatively charged functional groups. Similarly, analytes with positive charge or polarity are separated using a chromatographic support that carries positive charges. Any suitable detector can be used for the detection and quantitation of analytes in IC. The choice depends upon the nature of the analyte molecules. However, traditionally, IC is associated with electrochemical detectors.
Introduction
Two types of electrochemical detectors are widely used in IC—conductivity suppressed and nonsuppressed and pulsed amperometry. When a constant voltage is applied across two electrodes between which the effluent from a column flows, a current is generated because the effluent contains ions or polar molecules. The problem, however, is that the conductivities of mobile phases are significantly higher than the conductivities of the analytes, simply because the concentrations of ions in the former solutions are 10 4 5 higher than that of the analytes.
The suppressed conductivity detection permits detection and quantitation of analytes at near zero background baseline conductivity of the mobile phases. Used typically in combination with high-performance anion-exchange chromatography HPAEC, originally introduced as high-pH anion-exchange chromatography , Pulsed Amperometry Detection PAD has proved to be a powerful tool in the detection of mono- and oligosaccharides, sugar alcohols, aminoglycosides, amino acids and other molecules that do not have a suitable chromophore, without requiring any sample derivatization.
Since , ion exchange chromatography rapidly manifested into one of the most heavily leveraged techniques, with its principles often being applied to majority of fields of chemistry, including distillation, adsorption, and filtration. Ion-exchange chromatography separates molecules based on their respective charged groups.
Ion-exchange chromatography retains analyte molecules on the column based on coulombic ionic interactions. The ion exchange chromatography matrix consists of positively and negatively charged ions. The stationary phase consists of an immobile matrix that contains charged ionizable functional groups or ligands.
To achieve electroneutrality, these inert charges couple with exchangeable counterions in the solution. Ionizable molecules that are to be purified compete with these exchangeable counterions for binding to the immobilized charges on the stationary phase. These ionizable molecules are retained or eluted based on their charge.
Initially, molecules that do not bind or bind weakly to the stationary phase are first to wash away. Altered conditions are needed for the elution of the molecules that bind to the stationary phase. The concentration of the exchangeable counterions, which competes with the molecules for binding, can be increased or the pH can be changed. A change in pH affects the charge on the particular molecules and, therefore, alters binding.
The molecules then start eluting out based on the changes in their charges from the adjustments.
Pharma & Biopharma Resource Library | Thermo Fisher Scientific - US
Further such adjustments can be used to release the protein of interest. Additionally, concentration of counterions can be gradually varied to separate ionized molecules. This type of elution is called gradient elution. On the other hand, step elution can be used in which the concentration of counterions are varied in one step. Positively charged molecules bind to cation exchange resins while negatively charged molecules bind to anion exchange resins.
Cation exchange chromatography retains positively charged cations because the stationary phase displays a negatively charged functional group:. Before ion-exchange chromatography can be initiated, it must be equilibrated. The stationary phase must be equilibrated to certain requirements that depend on the experiment that you are working with. Once equilibrated, the charged ions in the stationary phase will be attached to its opposite charged exchangeable ions. Next, a buffer should be chosen in which the desired protein can bind to.
Search all learning center resources
After equilibration, the column needs to be washed. The washing phase will help elute out all impurities that does not bind to the matrix while the protein of interest remains bounded.
- Cheat Sheet: The 4-hour Workweek ...In 2 Minutes - The Entrepreneur’s Summary of Timothy Ferrisss Best Selling Book: (Updated and Revised).
- Less of a Stranger;
- Crystal Quest Part 5.
- Pharma & Biopharma Resource Library | Thermo Fisher Scientific - US.
- Who Shall Rule This American Nation??
This sample buffer needs to have the same pH as the buffer used for equilibration to help bind the desired proteins. Uncharged proteins will be eluted out of the column at a similar speed of the buffer flowing through the column.
Ion chromatography
Once the sample has been loaded onto to the column and the column has been washed with the buffer to elute out all non-desired proteins, elution is carried out to elute the desired proteins that are bound to the matrix. Bound proteins are eluted out by utilizing a gradient of linearly increasing salt concentration. With increasing ionic strength of the buffer, the salt ions will compete with the desired proteins in order to bind to charged groups on the surface of the medium. This will cause desired proteins to be eluted out of the column.
Proteins that have a low net charge will be eluted out first as the salt concentration increases causing the ionic strength to increase. Proteins with high net charge will need a higher ionic strength for them to be eluted out of the column. Thin layer chromatography or column chromatography share similarities in that they both act within the same governing principles; there is constant and frequent exchange of molecules as the mobile phase travels along the stationary phase.
It is not imperative to add the sample in minute volumes as the predetermined conditions for the exchange column have been chosen so that there will be strong interaction between the mobile and stationary phases. Furthermore, the mechanism of the elution process will cause a compartmentalization of the differing molecules based on their respective chemical characteristics. This phenomenon is due to an increase in salt concentrations at or near the top of the column, thereby displacing the molecules at that position, while molecules bound lower are released at a later point when the higher salt concentration reaches that area.
These principles are the reasons that ion exchange chromatography is an excellent candidate for initial chromatography steps in a complex purification procedure as it can quickly yield small volumes of target molecules regardless of a greater starting volume.
Comparatively simple devices are often used to apply counterions of increasing gradient to a chromatography column. Counterions such as copper II are chosen most often for effectively separating peptides and amino acids through complex formation. A simple device can be used to create a salt gradient. Elution buffer is consistently being drawn from the chamber into the mixing chamber, thereby altering its buffer concentration.
Generally, the buffer placed into the chamber is usually of high initial concentration, whereas the buffer placed into the stirred chamber is usually of low concentration. As the high concentration buffer from the left chamber is mixed and drawn into the column, the buffer concentration of the stirred column gradually increase.
Pharma & Biopharma Resource Library
Altering the shapes of the stirred chamber, as well as of the limit buffer, allows for the production of concave, linear, or convex gradients of counterion. A multitude of different mediums are used for the stationary phase. Successful packing of the column is an important aspect of ion chromatography. Stability and efficiency of a final column depends on packing methods, solvent used, and factors that affect mechanical properties of the column.
In contrast to early inefficient dry- packing methods, wet slurry packing, in which particles that are suspended in an appropriate solvent are delivered into a column under pressure, shows significant improvement. Three different approaches can be employed in performing wet slurry packing: Polystyrene is used as a medium for ion- exchange. It is made from the polymerization of styrene with the use of divinylbenzene and benzoyl peroxide. Such exchangers form hydrophobic interactions with proteins which can be irreversible.
Due to this property, polystyrene ion exchangers are not suitable for protein separation. They are used on the other hand for the separation of small molecules in amino acid separation and removal of salt from water. Polystyrene ion exchangers with large pores can be used for the separation of protein but must be coated with a hydrophillic substance. Cellulose based medium can be used for the separation of large molecules as they contain large pores. Protein binding in this medium is high and has low hydrophobic character. DEAE is an anion exchange matrix that is produced from a positive side group of diethylaminoethyl bound to cellulose or Sephadex.
Agarose gel based medium contain large pores as well but their substitution ability is lower in comparison to dextrans. The ability of the medium to swell in liquid is based on the cross-linking of these substances, the pH and the ion concentrations of the buffers used. Incorporation of high temperature and pressure allows a significant increase in the efficiency of ion chromatography, along with a decrease in time. Temperature has an influence of selectivity due to its effects on retention properties.
Despite ion selectivity in different mediums, further research is being done to perform ion exchange chromatography through the range of 40— o C. An appropriate solvent can be chosen based on observations of how column particles behave in a solvent. Using an optical microscope, one can easily distinguish a desirable dispersed state of slurry from aggregated particles. A "strong" ion exchanger will not lose the charge on its matrix once the column is equilibrated and so a wide range of pH buffers can be used.
If the pH of the buffer used for a weak ion exchange column goes out of the capacity range of the matrix, the column will lose its charge distribution and the molecule of interest may be lost. In some experiments, the retention times of weak ion exchangers are just long enough to obtain desired data at a high specificity. There are also special columns that have resins with amphoteric functional groups that can exchange both cations and anions. These two types of exchangers can maintain the charge density of their columns over a pH range of 5—9. In ion chromatography, the interaction of the solute ions and the stationary phase based on their charges determines which ions will bind and to what degree.
When the stationary phase features positive groups which attracts anions, it is called an anion exchanger; when there are negative groups on the stationary phase, cations are attracted and it is a cation exchanger. Each resin features relative selectivity which varies based on the solute ions present who will compete to bind to the resin group on the stationary phase. The selectivity coefficient, the equivalent to the equilibrium constant, is determined via a ratio of the concentrations between the resin and each ion, however, the general trend is that ion exchangers prefer binding to the ion with a higher charge, smaller hydrated radius, and higher polarizability, or the ability for the electron cloud of an ion to be disrupted by other charges.
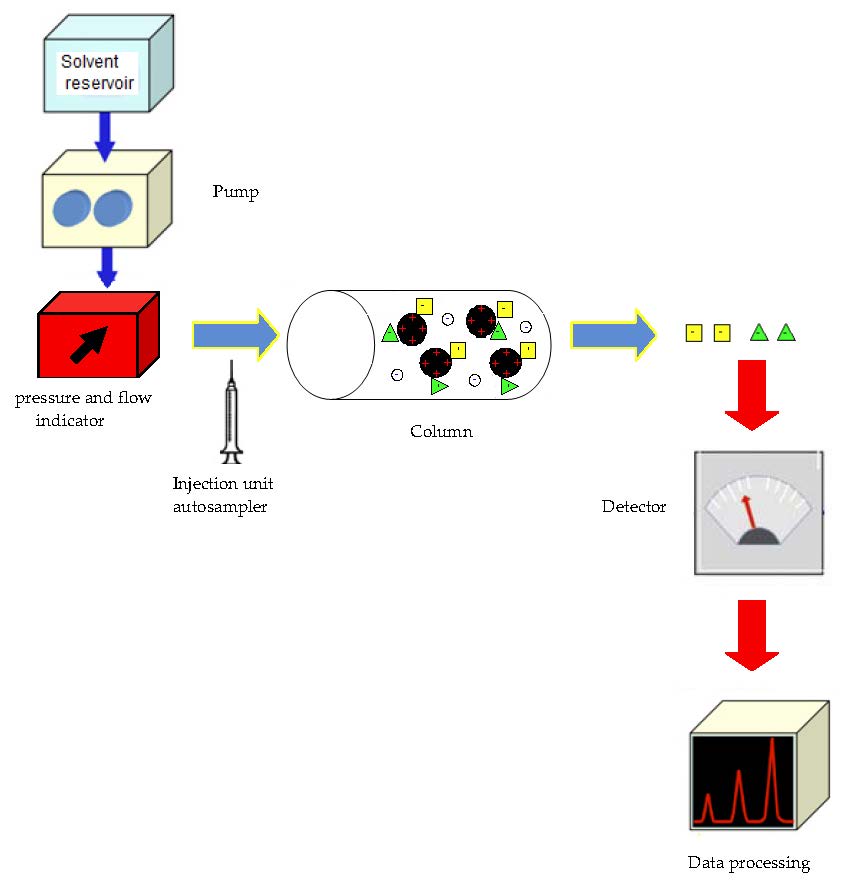
A sample is introduced, either manually or with an autosampler , into a sample loop of known volume. A buffered aqueous solution known as the mobile phase carries the sample from the loop onto a column that contains some form of stationary phase material. This is typically a resin or gel matrix consisting of agarose or cellulose beads with covalently bonded charged functional groups.
Equilibration of the stationary phase is needed in order to obtain the desired charge of the column. If the column is not properly equilibrated the desired molecule may not bind strongly to the column. The target analytes anions or cations are retained on the stationary phase but can be eluted by increasing the concentration of a similarly charged species that displaces the analyte ions from the stationary phase.
For example, in cation exchange chromatography, the positively charged analyte can be displaced by adding positively charged sodium ions. A type of ion exchange chromatography, membrane exchange [33] [34] is a relatively new method of purification designed to overcome limitations of using columns packed with beads. Membrane Chromatographic [35] [36] devices are cheap to mass-produce and disposable unlike other chromatography devices that require maintenance and time to revalidate. There are three types of membrane absorbers that are typically used when separating substances.
The three types are flat sheet, hollow fibre, and radial flow. The most common absorber and best suited for membrane chromatography is multiple flat sheets because it has more absorbent volume.
It can be used to overcome mass transfer limitations [37] and pressure drop, [38] making it especially advantageous for isolating and purifying viruses, plasmid DNA, and other large macromolecules. The column is packed with microporous membranes with internal pores which contain adsorptive moieties that can bind the target protein. Adsorptive membranes are available in a variety of geometries and chemistry which allows them to be used for purification and also fractionation, concentration, and clarification in an efficiency that is 10 fold that of using beads.
A more recent method involved the use of live cells that are attached to a support membrane and are used for identification and clarification of signaling molecules. Ion exchange chromatography can be used to separate proteins because they contain charged functional groups. The solutes are most commonly in a liquid phase, which tends to be water. Take for example proteins in water, which would be a liquid phase that is passed through a column. The column is commonly known as the solid phase since it is filled with porous synthetic particles that are of a particular charge.
These porous particles are also referred to as beads, may be aminated containing amino groups or have metal ions in order to have a charge. This is because slow diffusion of the solutes within the pores does not restrict the separation quality. The amino acids that have negatively charged side chains at pH 7 pH of water are glutamate and aspartate. The beads that are negatively charged are called cation exchange resins, as positively charged proteins will be attracted.
The amino acids that have positively charged side chains at pH 7 are lysine, histidine and asparagine. The isoelectric point is the pH at which a compound - in this case a protein - has no net charge. Using buffers instead of water for proteins that do not have a charge at pH 7, is a good idea as it enables the manipulation of pH to alter ionic interactions between the proteins and the beads.
Separation can be achieved based on the natural isoelectric point of the protein. Alternatively a peptide tag can be genetically added to the protein to give the protein an isoelectric point away from most natural proteins e. Elution by increasing ionic strength of the mobile phase is more subtle. It works because ions from the mobile phase interact with the immobilized ions on the stationary phase, thus "shielding" the stationary phase from the protein, and letting the protein elute.
Elution from ion-exchange columns can be sensitive to changes of a single charge- chromatofocusing. Ion-exchange chromatography is also useful in the isolation of specific multimeric protein assemblies, allowing purification of specific complexes according to both the number and the position of charged peptide tags. In ion exchange chromatography, the Gibbs—Donnan effect is observed when the pH of the applied buffer and the ion exchanger differ, even up to one pH unit.
For example, in anion-exchange columns, the ion exchangers repeal protons so the pH of the buffer near the column differs is higher than the rest of the solvent. This effect comes as a result of two similarly charged particles, one from the resin and one from the solution, failing to distribute properly between the two sides; there is a selective uptake of one ion over another. However, since the concentration of the sulphonic acid in the resin is high, the hydrogen of HCl has no tendency to enter the column.
This, combined with the need of electroneutrality, leads to a minimum amount of hydrogen and chlorine entering the resin. A use of ion chromatography can be seen in the argentation ion chromatography.